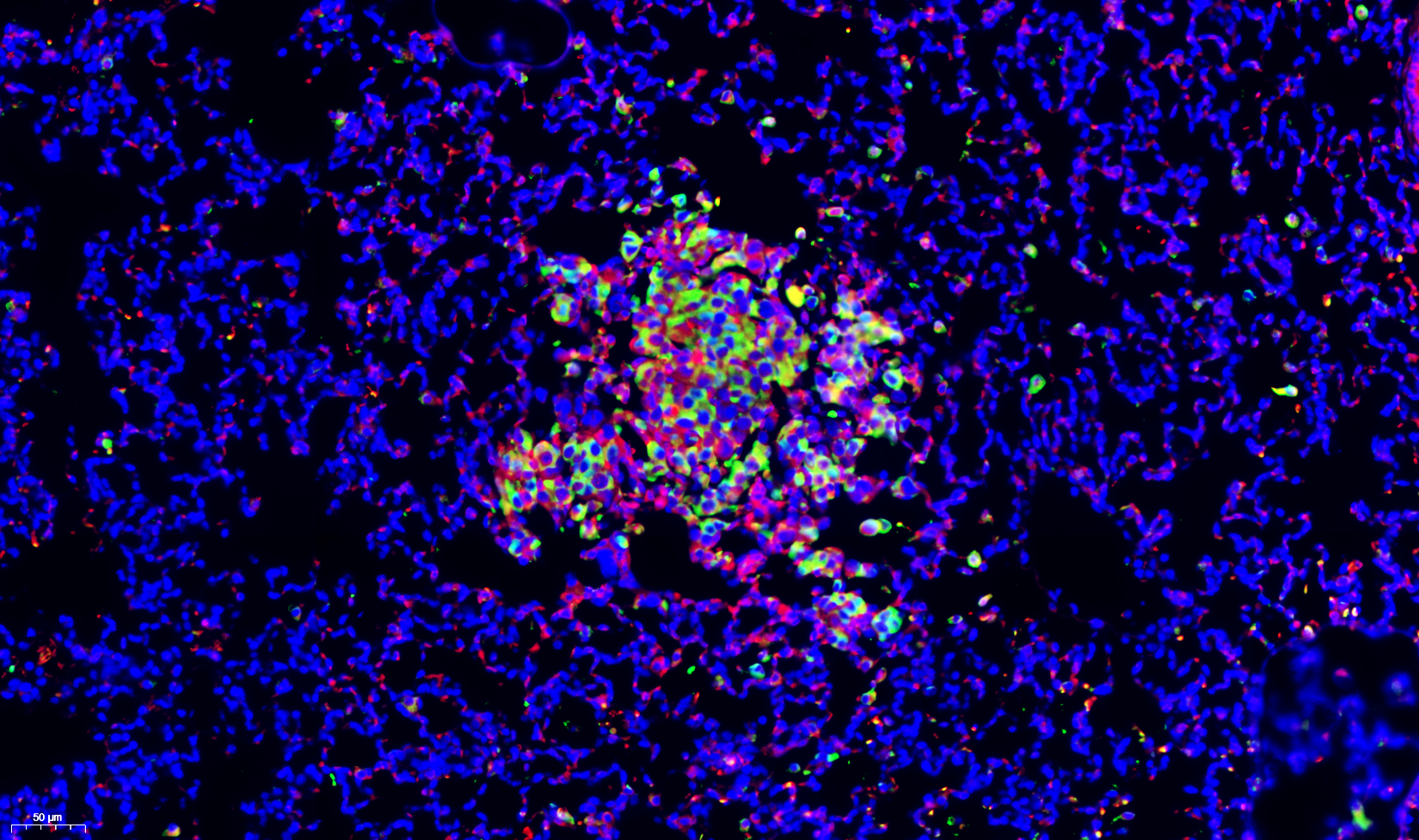
Research
Red: vasculature. Green: GFP-tumor cell. Cyan: Macrophages.
Source: https://www.nature.com/articles/s41467-022-28076-3
Our Research
We seek to understand the mechanisms of metastasis, with the goal of improving the lives of metastatic patients.
Metastasis can develop months, years, or even decades after the removal of the primary tumor. This is because tumor cells that have disseminated to distant organs do not grow immediately and survive in a non-proliferative state, called dormant state. Metastatic growth occurs when tumor cells exit dormancy (a process called “awakening”) and resume proliferation. As such, dormancy and metastatic recurrence are responsible for deaths of cancer patients.
Currently, there are no drugs to eradicate dormant tumor cells. This is because their biology is largely unknown. Therefore, our lab is interested in understanding how the tumor microenvironment regulates dormancy and awakening of tumor cells.
To this end, we use a multidisciplinary approach spanning multiplex immunofluorescence, organotypic culture, multiphoton intravital imaging, and in vivo models with the mission of developing novel therapeutic strategies to eradicate dormant tumor cells and combat mortality from metastasis.
Our lab focuses on two major questions:
1. How do tumor cells become dormant in the primary tumor?
Patient data and mouse models of different cancer types demonstrate that tumor cell dissemination occurs even before primary tumor is clinically detectable, and these early disseminated tumor cells (DTCs) are dormant and contribute to metastasis.These data suggest that the dormancy program initiates in the primary tumor and is carried over by DTCs in distant organs. However, it is unclear how the primary tumor instructs tumor cells to enter dormancy. We recently demonstrated that particular microenvironmental niches, rich in macrophages, in the primary tumor educate tumor cells for dormancy.
Our goal is to define the mechanisms by which macrophages and neutrophils induce dormancy.
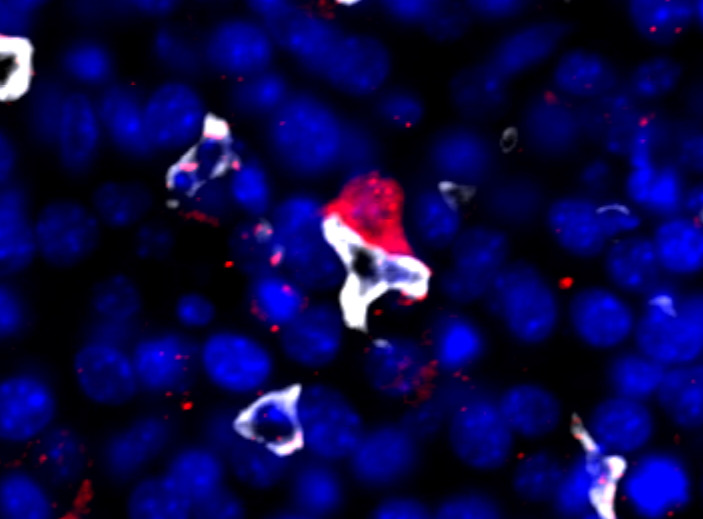
Source: https://www.nature.com/articles/s41467-022-28076-3
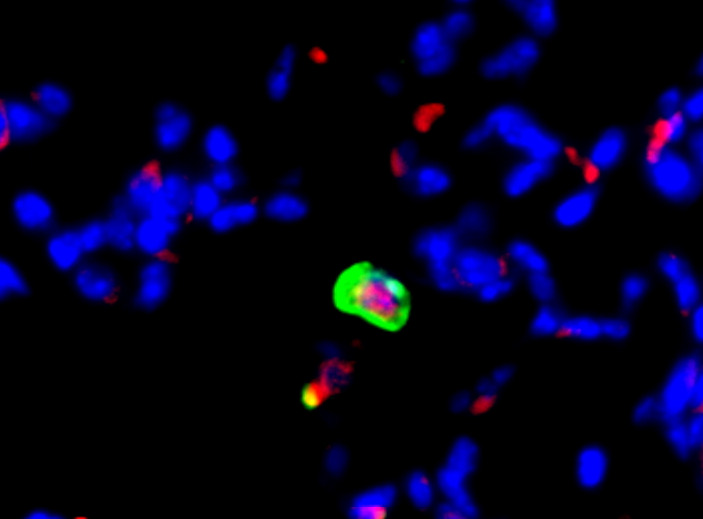
2. How do tumor cells exit dormancy at metastatic sites?
Tumor cells disseminate from the primary tumor and seed in distant organs, remaining dormant for many years. Upon unknown microenvironmental signals, DTCs exit dormancy and start to proliferate, leading to metastasis formation. However, the mechanisms and signaling that trigger DTCs to exit dormancy are poorly understood. The role of stromal cells in cancer progression and metastasis has been extensively demonstrated, but their contribution to the awakening of dormant DTCs has not been determined.
Our goal is to determine if fibroblast-derived soluble factors and extracellular vesicles induce DTCs to exit dormancy. The results of this project will identify new microenvironmental factors and signaling pathways that regulate awakening of dormant DTCs, and will provide new targets for therapeutic strategies to eliminate dormant cancer cells or to keep them in a perpetual dormant state.